Question: What makes a chemical a “vitamin C”?
Answer: Not just the name “ascorbyl” “ascorbate”
We find out that something is a vitamin WHEN it is missing. No vitamin, no health. In particular, deficiency of Vitamin C results in scurvy, and the name “Ascorbic Acid” is derived from the Latin word for scurvy (scorbutus), a nasty illness whose (easy) fix, citrus fruit, eluded pirates and sailors until 1753.
Why is L-Ascorbic Acid a vitamin? Most animals can make their own vitamin C, but humans can’t, because somewhere along evolution we lost a crucial enzyme, L-gulonolactone oxidase, required for the synthesis of L-Ascorbic Acid. This made L-ascorbic acid into an essential nutrient, i.e. we must get it by eating food containing it (or applying the vitamin to our skin).
 L-Ascorbic Acid is important for plants and animals because it works as an antioxidant. More specifically, it is a water soluble antioxidant, and this means that it can work in almost all the reactions occurring in the cell. [Conversely, chemicals with vitamin E activity are lipid (oil) soluble and can work in the lipid side of the cell membranes. They are both important, this is why they are vitamins!]
L-Ascorbic Acid is important for plants and animals because it works as an antioxidant. More specifically, it is a water soluble antioxidant, and this means that it can work in almost all the reactions occurring in the cell. [Conversely, chemicals with vitamin E activity are lipid (oil) soluble and can work in the lipid side of the cell membranes. They are both important, this is why they are vitamins!]However, there is a lot more to Vitamin C than “just” its antioxidant side. In humans and many animals ascorbic acid is also required for the synthesis of specific chemicals. For example, L-ascorbic acid is a cofactor in the synthesis of carnitine and tyrosine (an amino acid) and it is required for the synthesis of collagen, that very important protein in the skin.
Collagen is a protein of complex structure, and the final protein we require is very different from the peptides initially made at the ribosomes. Collagen is composed of a triple helix, which consists of two identical chains and an additional chain that differs slightly in its chemical composition. The amino acid composition of collagen is unusual for proteins, it has a high content of hydroxyproline. The peptides synthesized in the ribosomes undergo many modifications of their structure before they become collagen; among other modifications, proline (and lysine) residues in the peptides must be hydroxylated in a process catalyzed by enzymes that require ascorbic acid as a cofactor. The many symptoms of scurvy result from the inability of the human body to complete the transformation of the nascent peptides into collagen because of this lack of ascorbic acid. [Incidentally, here you see why it is silly to add hydroxyproline to a skin care product: this amino acid is NOT used in the synthesis of collagen. Proline is used and after protein synthesis, the proline residues are hydroxylated.]
Chemistry
- Vitamin C is a chemical, its chemical name is L-Ascorbic Acid.
- Why is L-Ascorbic Acid a vitamin? Most animals can make their own vitamin C, but humans can’t. We must get it by eating food containing it (or applying the vitamin to our skin).
- L-Ascorbic Acid is important for plants and animals because it works as an antioxidant, preventing damage to cells from oxidation reactions.
- L-Ascorbic Acid is also require to create collagen, an important protein in the skin.
- Un-oxidized L-Ascorbic Acid acts as a strong antioxidant; oxidized L-Ascorbic Acid acts as an oxidant and will be bad for the skin.
Effect
- Ascorbic Acid (L) provides important protection against damage induced by UV radiation (and the DNA mutations and cancer that may result from it), improves skin elasticity, decreases wrinkles by stimulating collagen synthesis, reduces redness, promotes wound healing and suppresses melanin synthesis.
- Visually, the use of L-Ascorbic Acid improves wrinkles, and decreases inflammation.
- Glycolic and lactic acids are frequently used in peels because they are weak acids that will do their job and later be metabolized by the skin. L-Ascorbic acid is also a AHA and useful for an acidic peel. [See below for information regarding the strength of Vitamin C Serums as an exfoliant]
- Chemical derivatives such as Magnesium Ascorbyl Phosphate (MAP) offer advantages such as stability and an acidity level that can make them more suitable for skin care products.
- MAP is a good vitamin C derivative, because it has been shown that it penetrates the skin and in the skin it is converted to L-ascorbic acid. As for most important question, is it an actual Vitamin C, MAP has been shown to protect the skin from UV damage and prevents synthesis of melanin, just as L-Ascorbic Acid does. This is why in our popular collagen serum you will find MAP and not L-Ascorbic acid.
- Sometimes an oil-soluble version of Vitamin C is wanted for a special formulation, and for that, we turn to Ascorbyl Palmitate. But it must be remembered that the principal role of vitamin C is as a water-soluble antioxidant.
- It is important not to combine Vitamin C with metallic ions such as Copper or Zinc in a formulation.
- A 20% concentration is pretty close to the maximum solubility and at that point you may see some crystals coming out of solution, this will occur more frequently in a refrigerated serum. If the Vitamin C comes out of solution, warm the serum up a little and shake well.
- It is very important that Vitamin C products contain un-oxidized L-Ascorbic Acid, old products with oxidized Vitamin C can cause damage to the skin.
- In non-acidic formulations, like our Collagen Serum, it is important to use a Vitamin C form like MAP or Ascorbyl Palmitate.
- At an acidic pH, L-Ascorbic Acid will be quite stable. The shelf life of the serum will depend on the acidity of the serum.
- Our new best estimate is that a serum with a pH. of 2.5 will have a shelf life of around 6 months.
- Because of the above point, we suggest checking the pH. of any L-Ascorbic Acid serum and ensuring that its pH. is below 3.5 (pH. strips can be purchased to measure this).
- Refrigeration slows the oxidation of L-Ascorbic Acid and extends the lifespan of the serum.
- Alpha Tocopherol and Ferulic Acid are added to L-Ascorbic Acid serums to delay the oxidation of Vitamin C.
- It is very important to make sure that you do not use oxidized L-Ascorbic Acid Serums. Make sure your product contains a ‘made on’ or ‘use by’ date. Do not use a product that is over 12 months old as it will not be useful to the skin. If mixing your own water/ascorbic acid mix do not keep the solution for more than a few hours. Refrigerate the products if possible.
- We recommend using a Vitamin C product daily, but for sensitive skin it is best to use a non-acidic form like MAP.
- High concentration L-Ascorbic Acid serums, or even fresh L-Ascorbic Acid crystals dissolved in water, are great exfoliating options.
- Because over exfoliation can cause inflammation and other problems we recommend careful use of L-Ascorbic Acid serums, once or twice a week is optimal for most skin types. Our Collagen Serum with MAP is a great alternative for daily use as it contains other valuable actives we cannot use in an acidic solution.
- Do not worry about waiting for the acidity of the skin to go back to normal (it will take a few hours) before using most other products. If you are using products with complex proteins, such as our EGF or our antioxidant products, it is recommended that you wait at least an hour to ensure the proteins are not damaged.
- DIY: Our ‘mix your own’ serum kit is optimal because freshness is assured, and the exfoliation level can be tailored to suit the user.
Questions and Answers
Question: What is the more stable form of Vitamin C?Answer: I assume your question refers to the oxidation of Vitamin C by oxygen. In this case, there are two parts to the answer
a) the ascorbic acid derivative must have Vitamin C activity in vivo (otherwise it is useless) and
b) are the stability measurements comparable across experiments?
I addressed the Vitamin C activity aspect in the main article (see above). The answer to b) is disappointing: there are plenty of good experiments made with L-ascorbic acid because of its medical importance. However, for commercially produced derivatives the quality of the data is not very good. For example, for ascorbyl tetraisopalmitate the evidence is sketchy (in my eyes, insufficient). Why? When the data are published in refereed scientific journals, the most important part of the paper is detailed information on how the experiments were performed (Materials & Methods section), but this vital information is usually missing from trade publications. The problem is that once companies have invested time and money in the development of a “new Vitamin C” there is no incentive to find flaws in their creation. So the experiments may be conducted only in vitro, or in conditions of pH and temperature that promote stability. These experiments are simply not useful.
Question: What is the best way to ensure that your vitamin C serum is fresh and has not oxidized?
Answer: The best way is to send it to analyze by a laboratory that has access to EPR (electron paramagnetic resonance). I know, that’s silly unless you are doing some serious scientific research. The second best way is to slow down oxidation by preparing the serum fresh, like when you use the Skin Actives kit. If all you are doing is adding ascorbic acid to water, all you need to do is throw it away when the color changes. The ascorbate ion is the predominant species at typical biological pH values. Upon oxidation, ascorbate is transformed and the colorless solution will turn brownish. This answer is also valid for commercially available C serums, like those sold by Peter Thomas Roth, Murad and Ole Henrikson. My suggestion is that you make your own C serum or, even better, use Skin Actives version. Why? We give you the double benefit of starting with L-ascorbic acid crystals (zero oxidation) and perfect formulation of the base serum, delaying by months any unwanted changes in the serum composition. In our serum, even if eventually you observe some changes in color, these are unlikely to be related to oxidation of ascorbic acid.
Question: How long should we wait before putting on serum or cream after using the Vit C serum?
Answer: Left to itself, the skin may take a few hours to go back near its original pH, i.e. if you don’t apply anything after the Vitamin C serum. But if you apply a different serum or a cream, the change back to near neutral pH will happen much sooner. I would not worry at all about the pH effect unless you wish to use serums containing delicate proteins, like EGF or thioredoxin.
Question:”How often should it be used if one’s regime already incorporates other serums and creams” and “The best regime for anti aging that incorporates it without compromising other ingredients. Vitamin C Serum For Dummies, if you will.”
Answer: It depends on your objectives, of course. But for older skin, I think (the acidic) ascorbic C serum could be used a couple of times a week, alternating with (the pH neutral) Collagen serum, which includes important active proteins like epidermal growth factor. And don’t forget Vitamin A, a retinoid, because it works in a completely different way and it is very useful in the treatment of wrinkles.
If you want to know more…. (stop when you get bored)
What does the “L-“ mean?
The L- in L-Ascorbic Acid refers to the position in space of the bonds between atoms (the broken lines in Figure 1). Many molecules have more than one possible structure in space because of the peculiar properties of the carbon atom. These are called asymmetric molecules and the old terminology allows for two mirror forms, L and D (levo and dextro, left and right in Latin, respectively) because of the effect a solution has on polarized light. L-Ascorbic Acid means that the compound’s stereochemistry is related to that of the levorotatory enantiomer of glyceraldehyde.
The Ascorbic Acid we use at Skin Actives Scientific is the L-Ascorbic form, but even if you were to get the racemic form (mix of the two stereoisomers) you would still be fine, because both forms have antioxidant activity, and you would get more than enough of the L-form to allow for the enzymatic reactions that require L-Ascorbic as a cofactor.
What can L-ascorbic acid do for your skin?
L-ascorbic acid is a water soluble antioxidant, a very important part of the antioxidant system that includes water-soluble and lipid soluble antioxidants and enzymes. In this complex system you will find many of the actives that we use at SAS: alpha-D-tocopherol, tocotrienols and Coenzyme Q10 (oil soluble), and glutathione (water soluble), superoxide dismutase and more (see our ROS* terminator).
How do they work together? When alpha-D-tocopherol donates an electron to “save” a membrane lipid, it becomes oxidized. Ascorbic acid re-reduces the oxidized vitamin E. The oxidized vitamin C is, in turn, re-reduced by glutathione and enzymes. This is the way that the system keeps working, recycling the oxidized antioxidants.
In short, vitamin C is the water soluble antioxidant that will decrease oxidative stress in your skin. This is probably the mechanism by which it inhibits melanogenesis, protects from UVA and UVB radiation, lightens sun spots and alleviates melasma.
The other effects of ascorbic acid on the skin are probably related to its role as a cofactor in a number of enzymatic reactions. At the biochemical level, L-ascorbic acid promotes synthesis of new collagen, promotes expression of type 1 and type 4 collagens and ascorbic acid transporters and inhibits the activity of matrix metalloproteinases, enzymes capable of breaking down collagen and elastin. Visually, the use of L-ascorbic acid improves wrinkles, and decreases inflammation.
What are all those “ascorbyl something” you see in the ingredient lists of skin care products?
Magnesium-L-Ascorbyl-2-Phosphate (MAP)
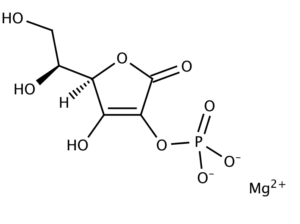
Figure: chemical structure of Magnesium-L-Ascorbyl-2-Phosphate
Dissolved in water, ascorbic acid may be destroyed by the oxygen in the air. This is not a problem when you are taking Vitamin C tablets or drinking orange juice, but it becomes a problem for skin care products that need to be on a store shelf for 12 months. Some products that contain ascorbic acid will change color and lose activity within weeks, making them useless.
Organic chemists have been playing with ascorbic Acid for a long time, trying to modify its structure to make it more suitable for use in skin care products, while keeping the Vitamin C activity intact. Every new chemical derivative requires testing to make sure that the modified molecule still works as Vitamin C in aspects relevant to the skin.Because each scientific paper measures activity in a different way it is important to read the scientific literature (and not just the marketing materials) to make sure that the chemically modified Ascorbic Acid is still useful.
Some product manufacturers insist that their modified ascorbic acids will be absorbed better than the natural form. This is a bad excuse for chemical modification because L-ascorbic acid is absorbed and used by the skin without any problem.
In the case of Magnesium-L-Ascorbyl-2-Phosphate (MAP), the L-Ascorbic Acid derivative we use in our Antioxidant and Collagen Serums, as the MAP crosses the epidermis it releases L-Ascorbic Acid. Ascorbyl 2-phosphates, usually formulated as the magnesium salts, are stable in solution at neutral pH (the phosphate group in the second position of the cyclic ring protects the enediol system of the molecule from oxidation). Ascorbyl phosphate salts are not in themselves antioxidant agents but that is not a problem, because in vivo they are converted into L-ascorbic acid [presumably accomplished by the enzyme alkaline phosphatase present in the skin].
MAP is a good vitamin C derivative, because it has been shown that it penetrates the skin and in the skin it is converted to L-ascorbic acid. As for most important question, is it an actual Vitamin C, MAP has been shown to protect the skin from UV damage and prevents synthesis of melanin, just as L-Ascorbic Acid does.
MAP offers an extra advantage to some users: for some, the acidity of an L-ascorbic acid solution can be a problem: it may sting. For the formulators, it allows Vitamin C to be formulated together with great skin actives like epidermal growth factor, which (as many other proteins) are unstable in acidic media. This is why in our popular (with good reason) collagen serum you will find MAP and not ascorbic acid.
Additionally, we also sell Ascorbyl Palmitate, another Vitamin C derivative. When mixing, in addition to the activity, you have to pay attention to solubility. Ascorbic Acid and Magnesium Ascorbyl Phosphate are water-soluble and therefore perfect for their Vitamin C and antioxidant properties required in our Collagen Serum and our Antioxidant Serum. However, sometimes an oil-soluble version of Vitamin C is wanted for a special formulation, and for that, we turn to Ascorbyl Palmitate. But it must be remembered that the principal role of vitamin C is as a water-soluble antioxidant.
Ascorbic acid as an alpha hydroxyl acid (AHA)
Glycolic and lactic acids are frequently used in peels because they are weak acids that will do their job and later be metabolized by the skin. Ascorbic acid is also a AHA and useful for an acidic peel.
Like with all acidic peels, the peel depth is determined by the pH (the lower the pH the more acidic and strong the peel), determined in turn by the type of acid, its concentration and whether it was neutralized. Volume applied, time of contact, frequency of application, integrity of skin, skin thickness, oiliness and post-peel care will also affect the results of the peel. AHAs in general will increase the production of collagen, water content of the skin, and synthesis of glycosaminoglycans. Acid peels will increase elastin fibers, volume of the epidermis, and tightening of the superficial skin layers. The lower the pH the stronger the effect but there may be more skin irritation.
In view of these facts, you can see that C serums will also work as mild peels, on top of the many benefits of Vitamin C as enzyme cofactor and antioxidant..
When is a C-serum NOT a C-serum?
Other Ascorbic Acid derivatives used in the industry include Ascorbyl Tetraisopalmitate (Tetrahexyldecyl Ascorbate), Ascorbyl Glucoside, and many more to come so that the marketing department can announce “the newest and most powerful vitamin C ever!” In this case, as in many others, new is not necessarily better, and we prefer to use MAP and Ascorbyl Palmitate, proven, stable forms of Vitamin C.
In short: For a C-serum to be actually a C-serum, the ascorbic acid derivative must be stable (so it is not oxidized before you apply to your skin), and it must have vitamin C activity in vivo. This will require that the derivative is broken down in the skin to produce ascorbic acid and has to display antioxidant activity, melanin inhibition, protection from UV radiation, etc. According to this very common sense definition, some C-serums in the market are probably NOT C-serums.
Other aspects of C serums
Why the recommended separation in time between using copper and vitamin C?
Ascorbic Acid has been shown to reduce copper (and iron) ions during the conversion from ascorbate to dehydroascorbate, a chemical reaction that can generate strong oxidants. There is not much free copper around in our bodies (it is bound to proteins) but for people who use Copper Peptide frequently, the interaction with vitamin C could become a problem. Although Copper Peptide helps with healing, when it comes to collagen, Vitamin C is far more important. Indeed, excess copper can inhibit collagen synthesis and promote its degradation. It is worth noting that having EDTA(used in some commercial C serums) around will not help because the metal is still available to ascorbic acid, a small molecule.
Why is pH so important?
Ascorbic acid is a diacid, i.e. it has 2 protons it potentially release to the medium.
What happens to ascorbic acid in solution? It depends on the pH. At pH 7.4, 99.95% of vitamin C will be present as AscH– (the form in the middle),0.05% as AscH2 and 0.004% as Asc2–. And what happens to AscH– in solution? It is a great antioxidant BUT it has a tendency to “fall apart”. The antioxidant form of Vitamin C, AscH– is also the “fragile” form. We can use the chemistry of L-ascorbic acid to our advantage. At an acidic pH, ascorbic acid will not work as an antioxidant, but it will also be stable. SAS C serum contains a stable form of ascorbic acid, the fully protonated form. Once applied to the skin, the serum will be absorbed into the skin and there, as the pH increases, ascorbic acid will work as an antioxidant and will do the other jobs of Vitamin C, no problem.
Refrigeration offers the benefit of slowing down all chemical reactions, so it is always beneficial to the stability of the serum overall.
Why are “C Serums” so popular?
Like for all skin care products, there is probably an important marketing component, but the relevance of Vitamin C to skin metabolism is so great that it is not surprising that Vitamin C serums actually do what they are supposed to do, a rarity in the skin care industry.
Ascorbic Acid (L) provides important protection against damage induced by UV radiation (and the DNA mutations and cancer that may result from it), improves skin elasticity, decreases wrinkles by stimulating collagen synthesis, reduces redness, promotes wound healing and suppresses melanin synthesis.
Ascorbic Acid increases collagen synthesis and the importance of collagen for skin health is paramount. Collagen fibers give the skin resistance to strain and traction. Collagen constitutes about 70% of skin mass, but total collagen decreases about 1% per year, what may look like a small decline, but in such a major component of the skin, it will affect skin volume and its physical properties. Also, aging changes collagen structure: what was an organized pattern in young skin, in older skin collagen can turn into thick fibrils arranged in disorganized bundles. It is not “only” quantity, then, it is also quality that matters. We know that aging decreases skin thickness and elasticity, and it is likely that collagen is a good part of the answer. If we care about slowing down and reversing skin aging, we should care about collagen too.
The price you pay for a long shelf life…
One of the most popular C serums on the market is C+E Skinceuticals . Here is the ingredient list:
Water, Ethoxydiglycol (a.k.a. diethylene glycol monethyl ether, solvent), Ascorbic Acid (L), Propylene Glycol (solvent), Glycerin, Laureth-23 (surfactant), Alpha Tocopherol, Phenoxyethanol, Triethanolamine (to increase pH), Ferulic Acid, Panthenol, Sodium Hyaluronate.
Some people that care a lot about ingredients don’t seem to mind using solvents that are not particularly “nice” like these in the Skinceuticals serum. The ingredients are required to keep the serum free of bacteria and mold, and to dissolve the Ferulic Acid. Alpha Tocopherol and Ferulic Acid are added to delay somewhat the oxidation of L-Ascorbic Acid. Even with these additions, many consumers will be disappointed to find that the serum color and smell are not “right”.
References:
Kameyama K, Sakai C, Kondoh S, Yonemoto K, Nishiyama S, Tagawa M, Murata T, Ohnuma T, Quigley J, Dorsky A, Bucks D, Blanock K. (1996) Inhibitory effect of magnesium L-ascorbyl-2-phosphate on melanogenesis in vitro and in vivo. J Am Acad Dermatol. 34:29-33.
Kobayashi S, Takehana M, Itoh S, Ogata E. (1996) Protective effect of magnesium-L-ascorbyl-2 phosphate against skin damage induced by UVB irradiation. Photochem Photobiol. 1996 Jul; 64:224-8
Geesin JC, Gordon JS, Berg RA. (1993) Regulation of collagen synthesis in human dermal fibroblasts by the sodium and magnesium salts of ascorbyl-2-phosphate. Skin Pharmacol. 6:65-71.
Claims on this page have not been evaluated by the FDA and are not intended to diagnose, cure, treat or prevent any disease.
